In this case, Δ G will be negative if the magnitude of the TΔ S term is less than Δ H. This condition describes an exothermic process that involves a decrease in system entropy. Such a process is spontaneous at high temperatures and nonspontaneous at low temperatures. If the TΔ S term is less than Δ H, the free energy change will be positive. In this case, Δ G will be negative if the magnitude of the TΔ S term is greater than Δ H. This condition describes an endothermic process that involves an increase in system entropy. Four possibilities therefore exist with regard to the signs of the enthalpy and entropy changes: Since T is the absolute (kelvin) temperature, it can only have positive values. The spontaneity of a process, as reflected in the arithmetic sign of its free energy change, is then determined by the signs of the enthalpy and entropy changes and, in some cases, the absolute temperature. One method involves the use of standard enthalpies and entropies to compute standard free energy changes, Δ G°, according to the following relation. A convenient and common approach to the calculation of free energy changes for physical and chemical reactions is by use of widely available compilations of standard state thermodynamic data. Calculating Free Energy Changeįree energy is a state function, so its value depends only on the conditions of the initial and final states of the system. Similar reasoning may be applied to a nonspontaneous process, for which the free energy change represents the minimum amount of work that must be done on the system to carry out the process. 
In addition, the technologies used to extract work from a spontaneous process (e.g., batteries) are never 100% efficient, and so the work done by these processes is always less than the theoretical maximum. However, as noted previously in this chapter, such conditions are not realistic. Where w max w max refers to all types of work except expansion (pressure-volume) work. This new property is called the Gibbs free energy ( G) (or simply the free energy), and it is defined in terms of a system’s enthalpy and entropy as the following: An alternative approach involving a new thermodynamic property defined in terms of system properties only was introduced in the late nineteenth century by American mathematician Josiah Willard Gibbs. One of the challenges of using the second law of thermodynamics to determine if a process is spontaneous is that it requires measurements of the entropy change for the system and the entropy change for the surroundings. Relate standard free energy changes to equilibrium constants.Explain how temperature affects the spontaneity of some processes.Calculate free energy change for a process using enthalpies of formation and the entropies for its reactants and products.Calculate free energy change for a process using free energies of formation for its reactants and products.Define Gibbs free energy, and describe its relation to spontaneity.Although it isn't trivial in general, you can check how the formula simplifies for processes mentioned below.By the end of this section, you will be able to: The general formula for work done by the gas is expressed as ∫p(V)dV if we consider pressure as the function of volume. These parameters in real gases differ from theoretical ones, but we already contain them in our thermodynamic processes calculator. R for gases with more complex molecules.ΔT, where Cv is molar heat capacity under constant volume. 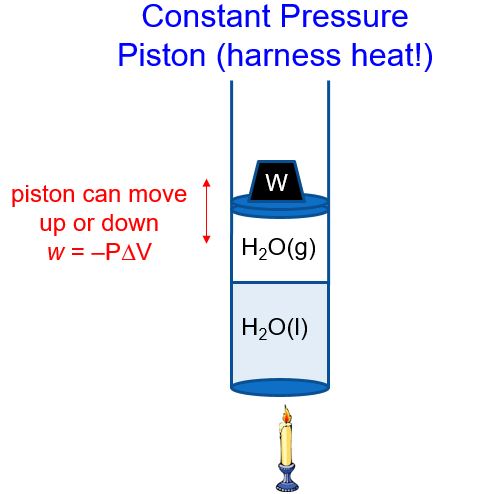
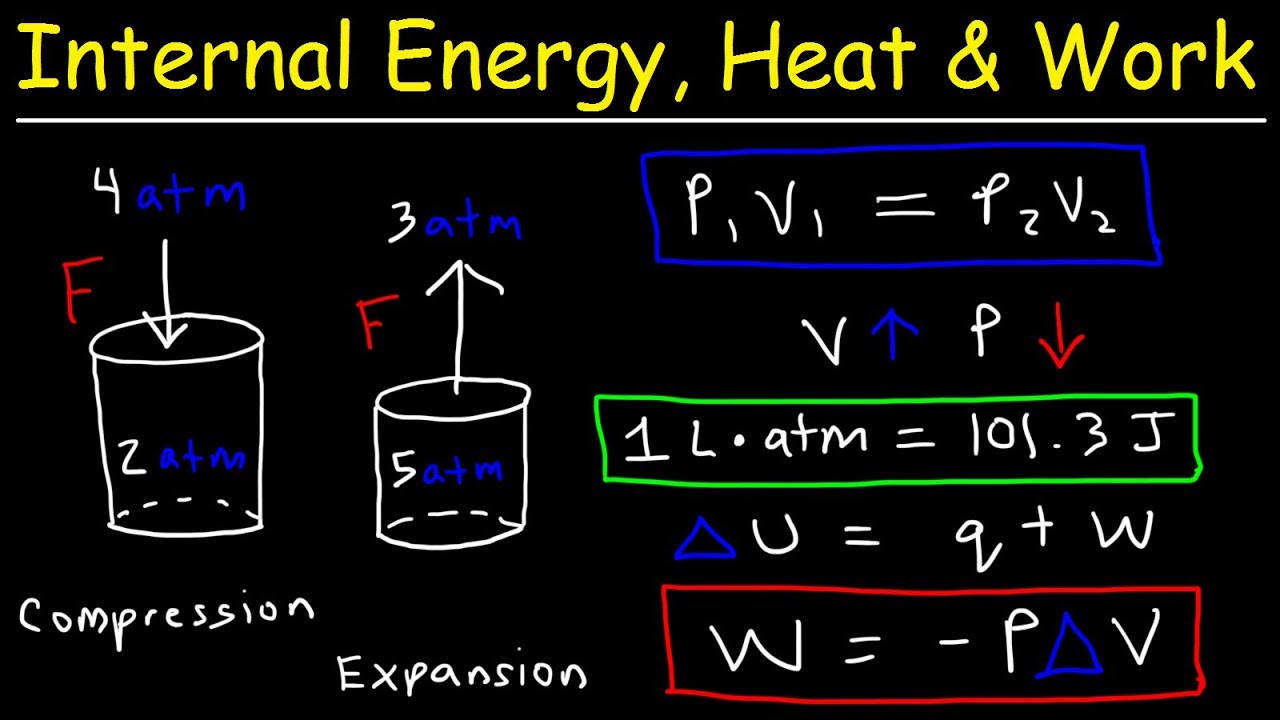
Internal energy change is proportional to temperature variation ΔT and type of gas with the following equation: ΔU = Cv Still, it is possible to find thermal energy changes ΔU, which are described by the first law of thermodynamics: ΔU = Q - W, where Q denotes heat absorbed, and W is work done by gas. It's quite tricky to estimate the precise value of internal energy. Internal energy U is the sum of all kinds of energy present in a system.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
